Abstract
Standard treatment of children and AYAs with LR first relapse of B-ALL [LR defined as bone marrow with or without extramedullary (BM±EM) relapse ≥36 months or isolated EM (IEM) relapse ≥18 months from initial diagnosis, and low (<0.1%) BM minimal residual disease (MRD) at the end of reinduction chemotherapy] consists of approximately 2 years of standard chemotherapy without hematopoietic stem cell transplant. The objective of this study was to compare survival [primary: disease-free (DFS); secondary: overall (OS)] of LR first relapse B-ALL patients aged 1-30 years randomized following reinduction chemotherapy (Block 1 of UKALLR3/mitoxantrone arm) to receive either two intensive chemotherapy blocks (Blocks 2 and 3 of UKALLR3) followed by continuation and maintenance chemotherapy of UKALLR3 (chemotherapy control) vs. the same except with integration of three 4-week cycles of blinatumomab, one replacing Block 3 chemotherapy and two added during continuation and maintenance (blinatumomab experimental). All patients with central nervous system (CNS) leukemia at relapse (isolated or combined with BM relapse) received additional intensified CNS-directed chemotherapy (intrathecal and systemic) and 1800 cGy of cranial radiation during maintenance. Patients with testicular leukemia at relapse that persisted after Block 1 reinduction received 2400 cGy testicular radiation during Block 2.
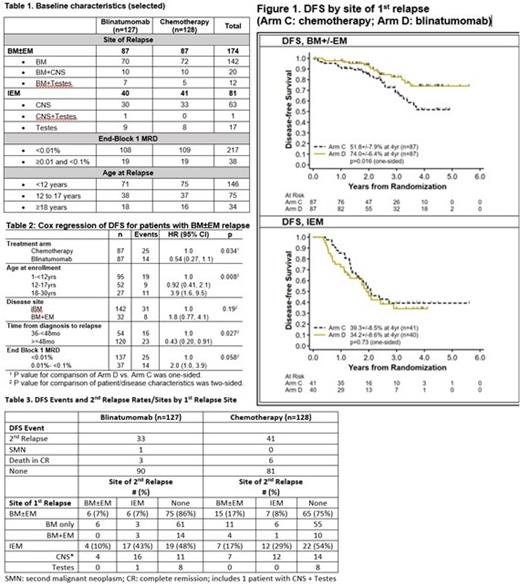
A total of 255 LR patients were randomized: Blinatumomab: 127; Chemotherapy: 128. Selected baseline characteristics are shown in Table 1. With median follow up of 2.9 years (data cut-off 12/31/20), the intent-to-treat (ITT) 4-year DFS (%±standard error) was 61.2±5.5% for blinatumomab vs. 48.2±6.0% for chemotherapy (p=0.15, 1-sided stratified log-rank test per pre-specified statistical plan). The 4-year OS was 91.6±3.0% for blinatumomab vs. 83.3±4.5% for chemotherapy (p=0.096).
Striking differences in DFS and blinatumomab efficacy were noted according to site of first relapse (Figure 1). For BM±EM relapses, 4-year DFS was 74.0±6.4% for blinatumomab vs. 51.8±7.9% for chemotherapy (p=0.016), and 4-year OS was 96.6±2.5% for blinatumomab vs. 84.4±5.6% for chemotherapy (p=0.013). Significant predictors of DFS in Cox multivariable regression for BM±EM relapses included treatment arm, age at relapse, and time from diagnosis to first relapse (Table 2). For IEM relapses, 4-year DFS was 34.2±8.6% for blinatumomab vs. 39.3±8.5% for chemotherapy (p=0.73), and 4-year OS was 81.7±7.0% for blinatumomab vs. 80.8±7.2% for chemotherapy (p=0.61). The only predictor of DFS in IEM patients was site of first relapse [hazard ratio for testes vs. CNS 0.19 (0.04-0.87), p=0.015]. The difference in DFS between BM±EM and IEM patients was driven by excess of second relapse in isolated CNS relapse patients (Table 3). Of 64 CNS relapses, 39 (61%) had a second relapse, of which 28 (72%) were also isolated CNS, with no difference by treatment arm. Of the 191 remaining patients, 35 (18%) had a second relapse [13 (14%) blinatumomab (6 BM±EM, 7 IEM), 22 (23%) chemotherapy (15 BM±EM, 7 IEM)].
Blinatumomab cycle 1 was better tolerated than Block 3 chemotherapy, with lower rates of CTCAEv4 grade ≥3 febrile neutropenia (3% vs. 47%, p<0.001), infections (5% vs. 51%, p<0.001), anemia (12% vs. 57%, p<0.001) and mucositis (1% vs. 7%, p=0.018). The rate of selected blinatumomab-related adverse events (AEs) in blinatumomab cycles 1/2/3 (all grades) were: Cytokine release syndrome (CRS) 12%/7%/7%, seizure 3%/1%/3%; other neurotoxicity (e.g., cognitive disturbance, tremor, ataxia, dysarthria) 19%/9%/5%. All blinatumomab-related AEs were fully reversible.
In conclusion, for children and AYA patients with LR first relapse of B-ALL, while there was no significant difference in outcome for the entire population, the blinatumomab arm was superior to the standard chemotherapy arm for patients with BM±EM relapse, establishing this regimen as a new standard therapy for these patients. The blinatumomab arm was not superior in IEM relapse. Isolated CNS relapse patients had a strikingly high relapse rate on both arms; better treatments are urgently needed for this subset.
Brown: Novartis: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Kura: Membership on an entity's Board of Directors or advisory committees; KIte: Membership on an entity's Board of Directors or advisory committees. Borowitz: Amgen, Blueprint Medicines: Honoraria. Raetz: Pfizer: Research Funding; Celgene: Other: DSMB member. Gore: Amgen: Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Kura Oncology: Membership on an entity's Board of Directors or advisory committees; Mirati: Current equity holder in publicly-traded company; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; OnKure: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Roche/Genentech: Consultancy; Sanofi Paris: Current equity holder in publicly-traded company. Whitlock: Amgen; Jazz Pharmaceuticals: Honoraria; Novartis: Research Funding; Sobi Pharmaceuticals: Consultancy. Hunger: Amgen: Current equity holder in publicly-traded company. Loh: MediSix therapeutics: Membership on an entity's Board of Directors or advisory committees.
Blinatumomab, used as post-reinduction consolidation without regard to MRD